Nature’s Proton Highway: Unraveling Charge Transport Mechanisms in Phosphate-Based Systems
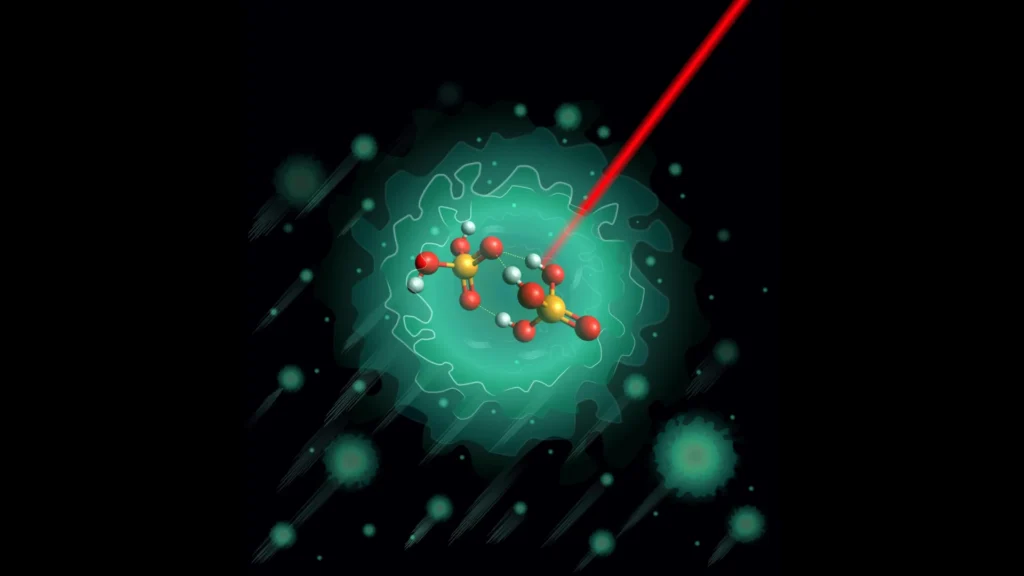
The movement of electrical charges within biological systems is fundamental to life, governing cellular communication, metabolism, and energy transfer. Among the molecules enabling this process, phosphoric acid and its derivatives play a central role due to their exceptional ability to conduct protons. This study explores the molecular mechanisms underlying proton transport in phosphate-based systems, revealing new insights into their structure and function through advanced experimental and theoretical approaches.
Introduction
Every second, billions of microscopic electrical charges traverse the human body, sustaining life by enabling biochemical communication and energy exchange. This charge transport is tightly regulated, particularly across cellular membranes, forming a fundamental control system in biological processes.
Phosphoric acid (H₃PO₄) and its related compounds are ubiquitous in living systems. They form the backbone of nucleic acids, contribute to membrane structure, and are integral to adenosine triphosphate (ATP)—the primary energy currency of cells. Beyond biology, their remarkable proton conductivity has made them indispensable in technological applications such as batteries and fuel cells.
Mechanism of Proton Transport
Unlike free-moving charged particles, protons in phosphate systems travel via a unique “proton-shuttling” mechanism. This involves hopping between molecules along hydrogen-bonded networks. These hydrogen bonds act as dynamic pathways, allowing rapid and efficient charge transfer.
Although the concept of proton-shuttling is well established, the precise molecular details governing the initial stages of proton transfer have remained elusive, necessitating deeper investigation.
Experimental Approach
To address this gap, researchers from the Department of Molecular Physics at the Fritz Haber Institute, in collaboration with international partners, examined a key molecular system: the deprotonated phosphoric acid dimer (H₃PO₄·H₂PO₄⁻).
The molecule was synthesized under controlled laboratory conditions and cooled to an extreme temperature of 0.37 Kelvin using helium nanodroplet isolation. This near absolute-zero environment minimized thermal disturbances, enabling highly precise structural analysis.
Infrared spectroscopy was employed to probe molecular vibrations, while quantum chemical calculations provided theoretical models of molecular behavior. The integration of these methods allowed for a comprehensive understanding of the system.
Results and Findings
Contrary to theoretical predictions suggesting the existence of two equally stable configurations, experimental observations revealed a single dominant structure.
This configuration is characterized by:
- A relatively rigid molecular framework
- Three hydrogen bonds interconnected through a shared oxygen atom
- High energy barriers that restrict internal proton movement
Such bonding patterns are consistent with those observed in other phosphoric acid clusters, indicating a potentially universal structural motif in phosphate-based systems.
Discussion
The discovery of a single stable structure challenges existing theoretical models and underscores the importance of experimental validation in molecular science. It suggests that current computational approaches may overlook subtle yet critical structural constraints.
Importantly, the rigidity and bonding arrangement identified in this study provide new insights into how proton conduction is regulated at the molecular level. Rather than unrestricted motion, proton transport appears to be governed by well-defined structural pathways.
Implications for Science and Technology
These findings have far-reaching implications across multiple domains:
- Biological Systems: Enhances understanding of proton transfer mechanisms in cellular processes, including energy metabolism and enzymatic activity.
- Materials Science: Provides a structural blueprint for designing advanced proton-conducting materials.
- Energy Technologies: Offers guidance for improving fuel cells and battery systems through optimized proton transport pathways.
- Theoretical Chemistry: Establishes a benchmark for refining quantum chemical models of hydrogen-bonded systems.
Conclusion
This research provides a significant step forward in decoding the molecular basis of proton transport in phosphate systems—often referred to as “Nature’s proton highway.” By identifying a single, well-defined structure for the key anionic dimer, the study bridges the gap between theoretical predictions and experimental reality.
Ultimately, these insights not only deepen our understanding of fundamental biological processes but also pave the way for innovations in energy and materials science, demonstrating the profound interconnectedness of life and technology.


